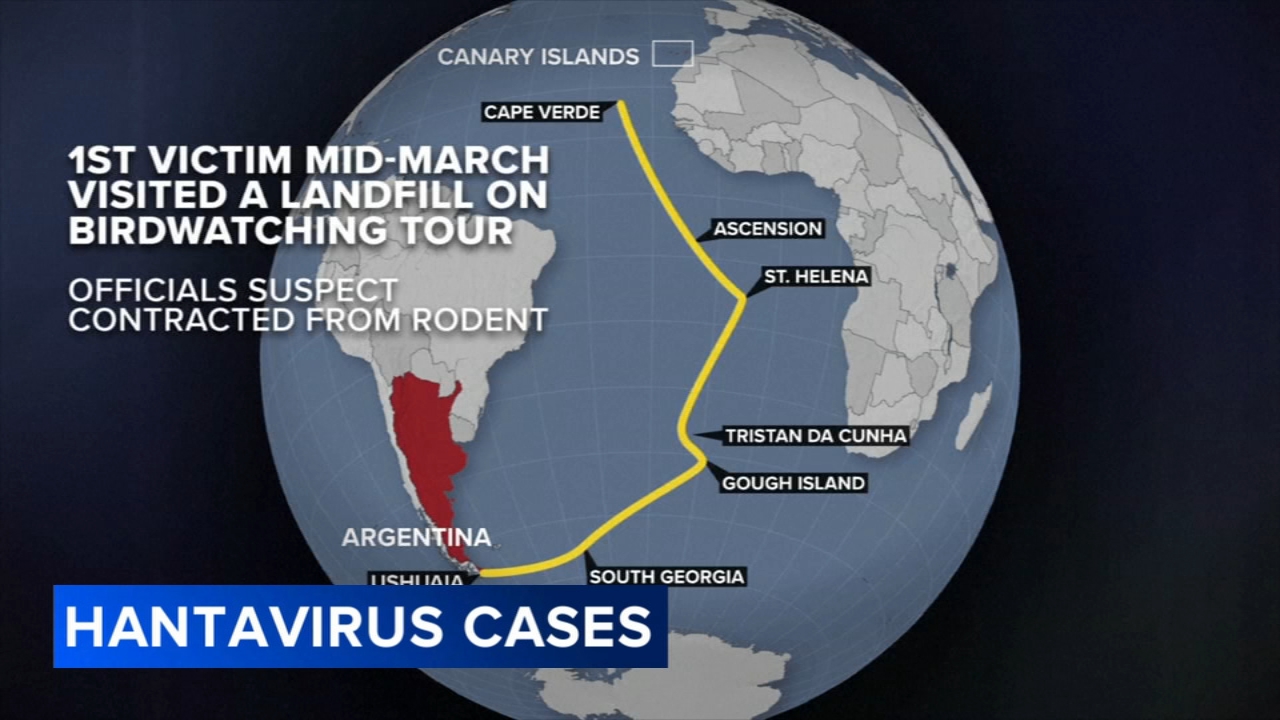
Pioneering mRNA technology in Moderna, Pfizer vaccines developed at University of Pennsylvania

PHILADELPHIA (WPVI) -- The pioneering work for the Moderna COVID-19 vaccine, which was granted emergency use authorization by the FDA, and the Pfizer vaccine began at the University of Pennsylvania more than a decade ago.
"This is an exceptionally safe vaccine; it's a shockingly effective vaccine." said Secretary of Health and Human Services Alex Azar.
On Friday night, the Moderna vaccine was overwhelmingly recommended by an FDA advisory panel for Americans 18 and up.
"By golly, the finish line is in sight. So we just got to keep on running. American people, we need you to keep on running." said U.S. Surgeon General Jerome Adams.
WATCH: Debunking COVID-19 vaccine rumors and myths
The FDA says approval of the Moderna vaccine follows a thorough evaluation of available safety, effectiveness and manufacturing quality information.
"We will likely see shots in the arm by the very early part of next week," Dr. Anthony Faucci of the Coronavirus Task Force said. "I would hope Monday or Tuesday."
RELATED: Penn Medicine researcher laid groundwork for Pfizer, Moderna vaccines

With the FDA's announcement, there is a big sense of pride for Dr. Drew Weissman, a professor of Infectious Diseases in the Perelman School of Medicine at the University of Pennsylvania.
His collaborative work with Dr. Katie Kariko, an adjunct associate professor at Penn and a senior vice president at BioNTech, beginning 15 years ago led to the development of these vaccines.

According to Penn Medicine, "[They] discovered in the early 2000s that introducing certain chemical modifications into messenger ribonucleic acid (mRNA) molecules can greatly increase its therapeutic potential-a discovery that plays a critical role in two of the leading COVID-19 vaccine candidates that rely on the use modified mRNA being separately developed by Moderna and Pfizer / BioNTech."
"My dream has always been to develop a drug, vaccine, treatment that helps people. This, I think, has accomplished that," said Dr. Weissman.
Unlike most vaccines which are a modified virus or viral protein to elicit an immune response, the Moderna and Pfizer vaccines use messenger RNA, mRNA, to instruct the body to begin defending itself against COVID-19.
"What's different about mRNA is it's a genetic vaccine," Dr. Weissman told Action News Health Reporter and Registered Nurse Ali Gorman last month.
Although Dr. Weissman says the differences between the two vaccines are very minor, we asked why the Pfizer vaccine requires storage in extreme cold temperatures and Moderna's does not.
"Moderna spent extra time developing and figuring out how to store it and how to prepare it so it could be stored at -20 instead of -80." said Dr. Weissman, who was also a fellow at Dr. Fauci's lab years ago.
The Moderna vaccine is expected to be delivered to states by Monday.
There are big hopes these two vaccines with others being developed could signal the beginning of the end of this historic pandemic.
Find out how many people may get a COVID-19 vaccine before you:
Interactive not displaying correctly? Click here to open in a new window.